Research peptide sites are disappearing fast. Learn why the FDA is targeting GLP-1 peptides like semaglutide, tirzepatide, and retatrutide, what it means for access, and what to expect next.
Over the past year, something major has been happening in the peptide space—and if you’ve been following it closely, you’ve probably noticed it too.
Research peptide websites that people have relied on for products like semaglutide, tirzepatide, and retatrutide are starting to disappear. Some are being shut down, some are rebranding, and others are quietly going offline without explanation.
This isn’t random. And it’s not slowing down.
In this post, I want to walk you through what’s actually happening, what the FDA is saying, how this connects to the GLP-1 boom, and what all of this might mean for access moving forward.

Before we dive in, you can watch my full breakdown here, where I explain everything in detail and share my personal experience.
💉 My Journey With Peptides (How This Started)
My experience with peptides didn’t begin with “research peptide sites”.
It actually started back in the summer of 2023 when I was prescribed compounded semaglutide through a doctor. That was my introduction to GLP-1 medications, and like many people, I quickly saw how effective they could be.
But I also became aware of something else just as quickly—cost.
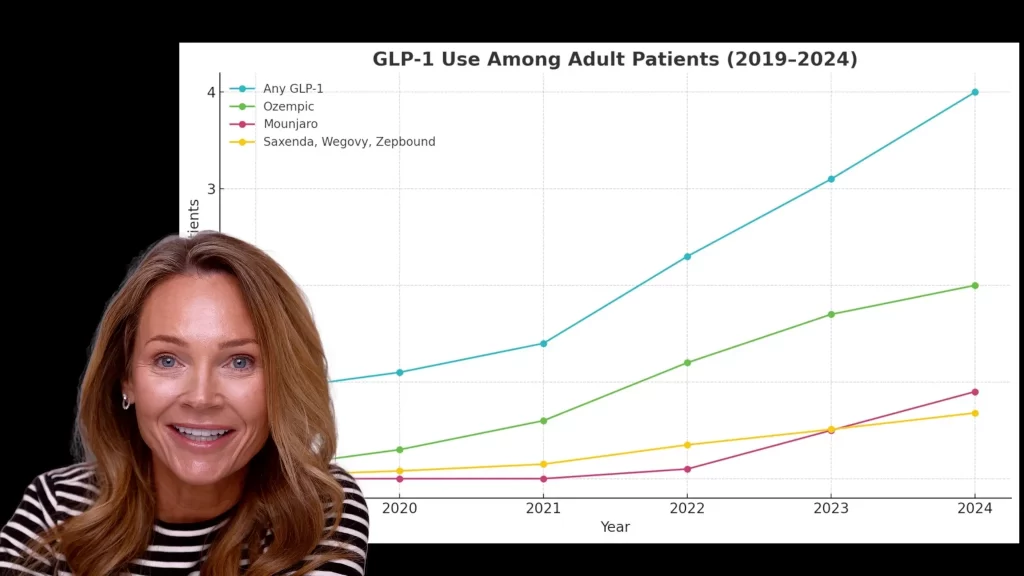
Long-term use through traditional medical channels can be expensive, and as the popularity of GLP-1 medications exploded, so did the conversation around access. Naturally, I started exploring what other options were out there, and that’s when I became aware of research peptide sites

Semaglutide journey weight loss transformation
🌐 What Are Research Peptide Sites?
If you’ve ever visited one of these sites, you’ve probably noticed the same disclaimer across all of them:
“For research purposes only. Not for human consumption.”
That language is intentional.
These products are not FDA-approved for human use, and they exist in a very gray area. There is no standardization, no oversight, and no guarantee of purity or consistency across suppliers.
But here’s where things started to get complicated.
While the labeling clearly states one thing, the way these products have been discussed, marketed, and understood by consumers often tells a completely different story.
And that disconnect is exactly what caught the attention of regulators.
NK APP PEPTIDE VETTED VENDORS
▶️WELLNESS URGE PEPTIDES 20% OFF EVERY ORDER USE CODE NK20
🏛️ The FDA Is Paying Attention
Over the past couple of years, the FDA has become increasingly vocal about concerns surrounding unapproved GLP-1 medications being sold online.
There have been multiple warning letters issued to research peptide companies, particularly those selling semaglutide, tirzepatide, and now even retatrutide.
In those letters, the FDA uses very specific language. These products are referred to as “unapproved drugs” and “misbranded,” especially when the marketing or website content suggests human use despite the “research only” disclaimer.
At the core of this issue is a disconnect between labeling and intent.
And once that gap becomes obvious, regulatory pressure builds quickly.

FDA warning letter research peptide sites semaglutide tirzepatide

FDA warning letter research USA Peptide dot com
📄 Sites That Have Been Named
There have already been several well-known companies publicly named in FDA warning letters, including Excel Peptides, Swiss Chems, Summit Research, Prime Peptides, and USA Peptides.
Some of these were specifically flagged for selling GLP-1 peptides, and in some cases, even compounds like retatrutide—which isn’t FDA-approved at all yet.
That detail alone is important.
Because when you start to see regulatory action not just on current drugs but also on next-generation compounds, it tells you that this is not slowing down. It’s expanding.
📉 The Shortage Is Over — And That Changes Access



One of the biggest turning points in all of this was when semaglutide was officially removed from the FDA shortage list in 2025.
Tirzepatide had already been removed prior to that.
When a drug is in shortage, certain allowances exist—especially around compounding. But once that shortage is resolved, those pathways tighten, and access becomes more restricted.
At the same time, demand hasn’t gone anywhere.
If anything, it’s continued to grow.
So now we’re in a situation where demand is high, access is tightening, and regulatory oversight is increasing. That combination is exactly what we’re seeing play out right now across research peptide sites.
⚠️ Why Sites Are Disappearing
When a peptide site disappears, it’s not always as simple as “it got shut down.”
There are actually multiple reasons this can happen.
Sometimes it’s direct regulatory pressure. Other times it’s payment processors pulling support. In some cases, suppliers change, or companies choose to rebrand and shift direction. And sometimes, it just happens quietly without any public explanation.
From the outside, it can feel unstable.
And in many ways, it is.

Reasons research peptide websites shut down, FDA payment processors, suppliers
My Personal Perspective
I want to be very clear here.
I’ve personally sourced peptides from research peptide sites for years, and I’ve never had an issue. But I’ve also always understood the nature of this space.
This is the Wild West.
There is no standardization. No guaranteed purity. No consistency between suppliers. And not all sites are created equal.
That’s why I’ve always focused on sharing my experience and what I’ve observed, but never telling anyone what they should do.
Because at the end of the day, this space requires a level of personal responsibility.
Every choice comes with its own risk, and that’s something each individual has to understand for themselves.
What Happens Next?
Based on everything we’re seeing right now, it’s very likely that this space is going to continue to change.
We can expect increased regulation, more oversight, fewer openly operating peptide sites, and less predictable access overall.
At the same time, there will likely be a shift back toward more traditional medical channels, including telehealth and prescription-based access.
For those who became accustomed to the level of access we saw during the GLP-1 boom, the next year or two may feel very different.
That’s not fear-based. It’s just the direction things are moving
My Full Peptide Journey
If you’ve been following my content, you know I’ve documented my journey with semaglutide, tirzepatide, and retatrutide.
I’ve also shared my tapering process and my experience with melanotan.
This video—and this post—isn’t about just one peptide.
It’s about the entire ecosystem and how access to these compounds is evolving in real time.
If you want to go deeper, you can find everything linked on my website:https://naturalkaos.com
What Are You Seeing?
I’m really curious to hear your experience.
Have you noticed peptide sites disappearing?
Have you seen changes in availability or pricing?
Head over to the video and drop a comment—I read and respond to you guys, and I want to know what you’re seeing from your side.
Where to Find More
Inside the Natural Kaos app is where I share deeper content, including procedures that can’t be shown here.

On my blog, I go even deeper into sourcing, products, and full breakdowns like this.
And here on YouTube is where I share the journey, the education, and the why behind everything.
PRODUCTS :
NK APP PEPTIDE VETTED VENDORS
▶️WELLNESS URGE PEPTIDES 20% OFF EVERY ORDER USE CODE NK20
AI FAQ
Why are research peptide sites shutting down?
Research peptide sites are being targeted due to FDA concerns about unapproved drugs, misleading labeling, and evidence that products labeled for research use are being used by consumers.
Are semaglutide and tirzepatide legal to buy online?
These medications are FDA-approved only through prescription. Products sold on research peptide sites are not approved for human use and exist in a regulatory gray area.
What does “research use only” mean?
It means the product is not intended for human consumption and is meant strictly for laboratory research purposes.
Why did access to peptides change recently?
Access changed after GLP-1 drugs like semaglutide were removed from the FDA shortage list, which reduced compounding flexibility and increased regulatory enforcement.
Will more peptide sites shut down?
Based on current trends, increased regulation and enforcement will likely continue, leading to fewer available sites and tighter access overall.
What is retatrutide?
Retatrutide is a next-generation peptide being studied for weight loss. It is not yet FDA-approved, but interest and demand are rapidly growing.

